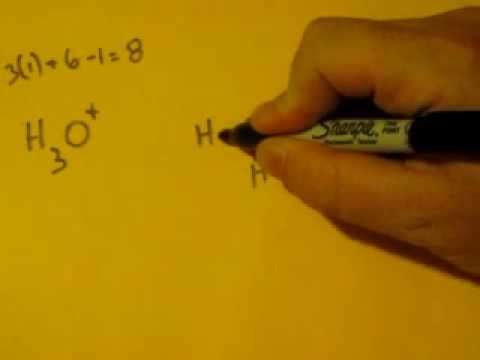
Where the only variable is r, the distance from the electron from the nucleus.
So both the orbitals and their energies, as displayed on the diagram are obtained from quantum theory. Note that there is a specific energy for each wave function or orbital.For example, f 1 might be the 1s AO, f 2, the 2s, etc. The f's are subscripted because there are many of them.Where the Greek letter f is a symbol for the mathematical function which represents the wave function or orbital.It is reasonable to ask "Where do we get these AO's and how do we know their shapes and sizes?" The answer is- we get them from quantum theory by solving the Schrodinger equation: The letter designating an AO indicates its shape, as noted before. The numeral designating an AO (e.g., the 1 in 1s) is a rough indication of orbital size, the smaller the number for a given atom, the smaller the orbital.Thus the IP is positive but the orbital energy is negative. This IP energy is equal to the orbital energy, but with opposite sign. Since the electron is stabilized by its attractive interaction with the positively charged nucleus, it requires energy to remove it. The ionization potential of an electron (IP) is the energy required to remove it from the nucleus entirely.
#HYDRONIUM ION BONDING AND HYBRIDIZATION FREE#
The AO energies are negative because the electrons in these AO's are attracted to the nucleus, making them more stable ( of lower energy) than free electrons. The energies of all AO's are negative with respect to the energy of an isolated, free electron at rest, which is taken as the zero of the energy scale.Orbitals are grouped into concentric shells of orbitals of comparable energy, e.g., the 1st, 2nd, or 3rd Mainshells, with only one AO (the 1s AO) in the first main shell, 4 in the 2nd main shell (2s,2p x, 2p y, and 2p z), 9 in the 3rd main shell, etc."s" atomic orbitals are spherically symmetric in shape, p orbitals are dumbbell shaped, etc.Electrons occupy orbitals (atomic orbitals, AO's), e.g., 1s,2s,2p, etc.which are centered around the nucleus.MOLECULAR STRUCTURE-The specific, three-dimensional arrangement of atoms in a molecule. A Reaction is the conversion of one molecule into another.ĪTOMIC STRUCTURE- The distribution of electrons in the space around a nucleus.Structure, in the case of a molecule, is the geometric description of a molecule, including such things as its shape, size, bond lengths, bond angles, and dihedral angles.For our purposes the "chemistry" being referred to here is the structure of these carbon compounds and their reactions.STUDY AIDS: (1) Emphasis Topics for each chapter are available on these web pages.(2 )Answer Keys for Last Year's Exams are also included in these web pages(3)The Instructor's Notes are also on these web pages(4) Problems in the textbook-as assigned in your syllabus (4)Review Sessions: To be announced (5) Office Hours (NLB and 2 TA's) (6) Office hours of other TA's involved in teaching the labs will soon be announced and are also available to you.ĭefinition: ORGANIC CHEMISTRY-The chemistry of the compounds of Carbon.SPECIAL URGENT ADVICE : This course very strongly emphasizes the lecture material and therefore has as its standard the mastery of this material to a very high level of excellence and detail.GOALS: (1) Develop rigorous, systematic reasoning processes valid in chemistry and related sciences (2) Develop understanding of chemistry (3) See the beauty and utility of chemistry as the "central science".
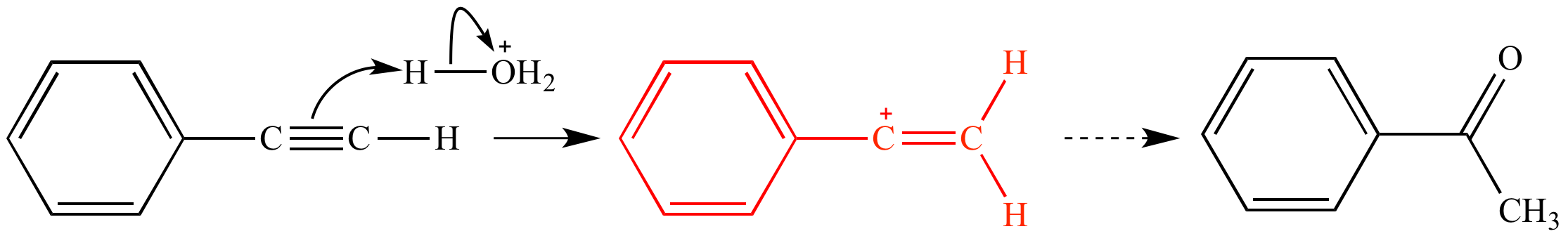
#HYDRONIUM ION BONDING AND HYBRIDIZATION HOW TO#
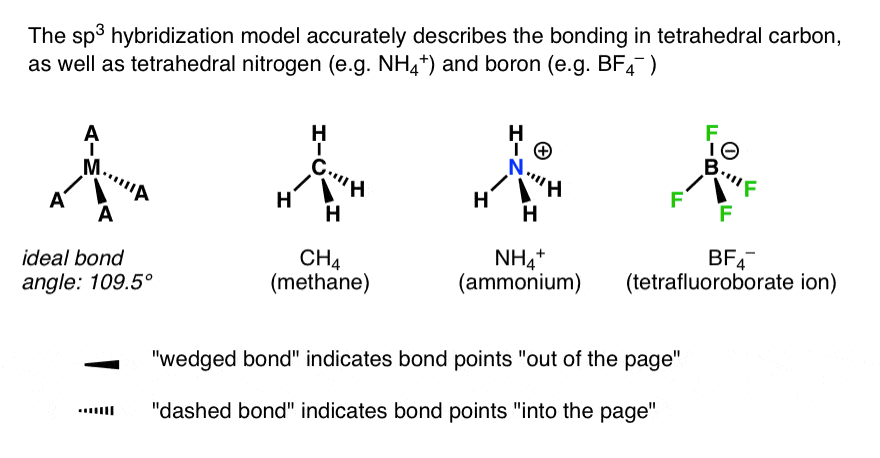
PERSONAL TEACHING EMPHASES (IN ORDER OF PRIORITY): (1)Reaction Mechanisms-Understanding how reactions occur at the molecular level (2)Theory-Understanding why reactions occur in the way they do (3) Synthesis-Understanding how to use reactions to convert one compound into another.Requirements_for_Strong_Covalent_Bonding.
